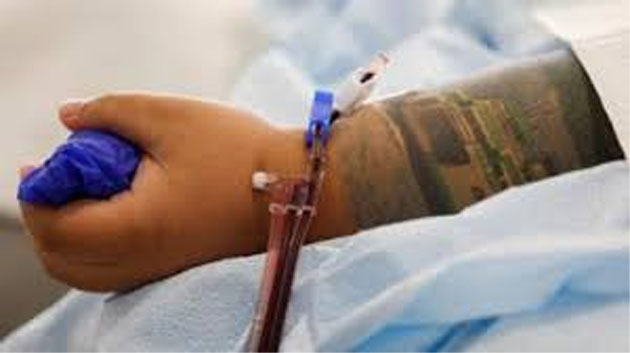
Washington, Aug 24:The US Food and Drug Administration (FDA) on Monday gave emergency authorisation for the use of plasma to treat coronavirus patients, a BBC report said.
The technique uses antibody-rich blood plasma from people who’ve recovered from the disease, and has already been used on over 70,000 people in the US.
President Donald Trump said the treatment could reduce deaths by nearly 35%.
This comes a day after he accused the FDA of impeding the rollout of vaccines and therapeutics for political reasons and on the eve of the Republican National Convention, where Mr Trump will launch his campaign for a bid for a second term in the White House.
“This is what I’ve been looking forward to doing for a long time,” the president told reporters. “I’m pleased to make a truly historic announcement in our battle against the China virus that will save countless lives.”
President Trump described the procedure as a powerful therapy and he appealed for Americans to come forward to donate plasma if they’ve recovered from COVID-19.
The FDA said early research suggests blood plasma can decrease mortality and improve patient health if it’s administered within the first three days of their hospitalisation.
The agency said it came to the conclusion about the safety of this treatment after an extensive review of data collected during recent months.
(UNI)